With the large-scale mining of gold, the easily leachable gold resources are gradually exhausted, and the refractory gold will become the main resource of gold industry in the future. According to statistics, about 1 / 3 of the world’s total gold output is produced from refractory gold mines. Among the proven gold reserves in China, 30% are difficult to deal with. According to the summary of many years of experience, Fodamon engineers have adopted the following pretreatment methods for refractory gold ores: roasting method, pressurized oxidation method, chemical oxidation method, etc., but they have the disadvantages of low gold recovery rate, large investment, large pollution and high cost of environmental protection control. Therefore, the pollution-free microbial oxidation technology is more attractive under the conditions of the decreasing rich and easily treated mineral resources, the continuous improvement of environmental protection requirements, and the increasing demand for metals in the development of modern industry and science and technology.

The so-called refractory gold ore refers to the ore and concentrate that can not obtain satisfactory leaching rate by direct extraction of gold by traditional cyanidation under normal grinding conditions. It is also called refractory gold ore or refractory gold ore. According to the different reasons for its difficulty, it can be divided into the following three categories.

(1) Wrapped gold deposit. In this kind of gold ore, gold is wrapped in arsenopyrite, pyrite, arsenopyrite, chalcopyrite, pyrrhotite, galena, sphalerite and other sulfide ores with quite fine particle size, which prevents the effective contact between gold and cyanide and hinders the leaching of gold. The particle size of this kind of gold ore is very small, so it is difficult to expose the gold particles by fine grinding or ultra-fine grinding. This kind of gold ore is usually directly cyanidated by conventional cyanidation, and the extraction rate is less than 40%.
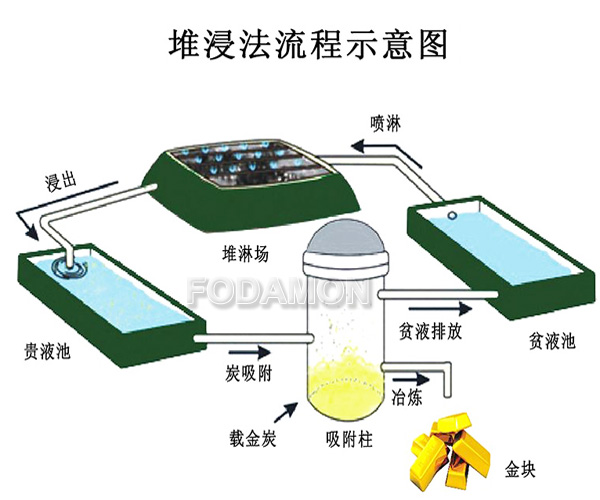
(2) Carbonaceous gold deposit. This kind of gold ore contains a certain amount of organic carbon and inorganic carbon. When extracting gold, in addition to the cyanidation process of gold into the solution, there is also the “gold robbing effect” caused by the adsorption of gold cyanide complexes in the solution by carbon substances, which makes the leached gold return to the leaching residue and affects the leaching of gold.
(3) Complex Polymetallic symbiotic gold deposit. Many gold mines are often symbiotic with sulfide minerals such as copper, zinc, antimony, mercury and tellurium and their oxide minerals. These complex polymetallic symbiotic mines are difficult to economically separate a single concentrate. During cyanidation, the symbiotic metal minerals are mostly unstable, and the sulfide and harmful impurities in the ore (usually exist in large quantities) will react with cyanide, oxygen or alkali, consuming a large amount of cyanide, oxygen and alkali, The cyanide products produced by the reaction often reduce the dissolution rate of gold after entering the solution; When the metal sulfide in the ore contacts with gold, it will lead to the dissolution and passivation of the gold anode. For this kind of ore, it is necessary to turn the conductive sulfide ore into a non-conductive oxide ore. some metal minerals such as gold telluride and black blunt gold ore, which are formed during reduction roasting, dissolve slowly or even insoluble in cyanide solution; Iron oxide, antimony, lead and other compounds in the ore will form a protective film on the surface of gold particles and hinder the leaching of gold. Generally, the gold ores suitable for bacterial oxidation pretreatment are mainly the first category, and the most common are arsenopyrite and pyrite gold ores.

To sum up, the reasons why these gold mines are difficult to deal with can be summarized as follows:
(1) Physical wrapping, mechanical wrapping, chemical crystal solid melt and chemical covering film, so that cyanide can not contact with gold ore;
(2) In the presence of cyanide and oxygen consuming substances, metal sulfides and oxides such as arsenic, copper, antimony, iron, manganese, nickel and cobalt have high solubility in the solution, and consume a large amount of cyanide and dissolved oxygen in the solution;
(3) The existence of gold robbing substances, such as carbonaceous substances, clay and other gold robbing substances, can adsorb gold complexes when leaching gold, and gold is “hijacked”;
(4) The existence of conductive minerals and some compounds formed by gold and conductive minerals such as tellurium, tin and antimony make the cathode of gold dissolved and passivated. Therefore, top treatment is generally required before leaching.
